
More Weight Control, Nutrition & Exercise Articles
The gut-brain connection: how your appetite is really controlled
Most people assume that hunger is simple. You get hungry, you eat, you stop. But if you have ever found yourself eating when you were not physically hungry, craving something specific even after a full meal, or struggling to stop after a few bites, you already know that appetite is far more complicated than that.
The science of hunger has changed considerably over the last two decades. Appetite is now understood as a complex biological system involving your gut, your hormones, your brain, and the nervous signals travelling between them. Willpower is part of the picture, but a relatively small part. Most of what drives your hunger and fullness is happening below the level of conscious awareness, in a biochemical conversation between your digestive system and your brain that never really stops.
Understanding how this system works not only explains why dieting is so hard, it also explains why newer approaches to weight management are producing results that traditional methods could not.

Appetite is regulated by hormonal signals your body generates automatically. Conscious control has less influence over hunger than most people believe.
The gut-brain axis: your body’s appetite highway
The gut-brain axis is the bidirectional communication network linking your gastrointestinal tract to your central nervous system. It operates through two channels working in parallel.
The first is the vagus nerve, a long nerve running from your brainstem all the way down to your digestive organs. This is the “wired” pathway: real-time neural feedback from your gut to your brain, operating continuously as you eat. When your stomach stretches, when food enters your small intestine, when nutrients hit specific sensors in your gut wall, the vagus nerve carries those signals upward to the brainstem and hypothalamus within seconds.
The second channel is hormonal, what researchers call the “wireless” system. Your gut, your fat tissue, and your pancreas all produce hormones that travel through the bloodstream to receptors in the brain. These hormonal signals regulate appetite over longer timeframes, shaping your overall drive to eat across hours and days rather than responding to the immediate sensation of a full stomach.
When both systems work properly, your appetite is roughly calibrated to your energy needs. When either system breaks down, as often happens in overweight and obesity, hunger becomes harder to regulate and fullness harder to feel.
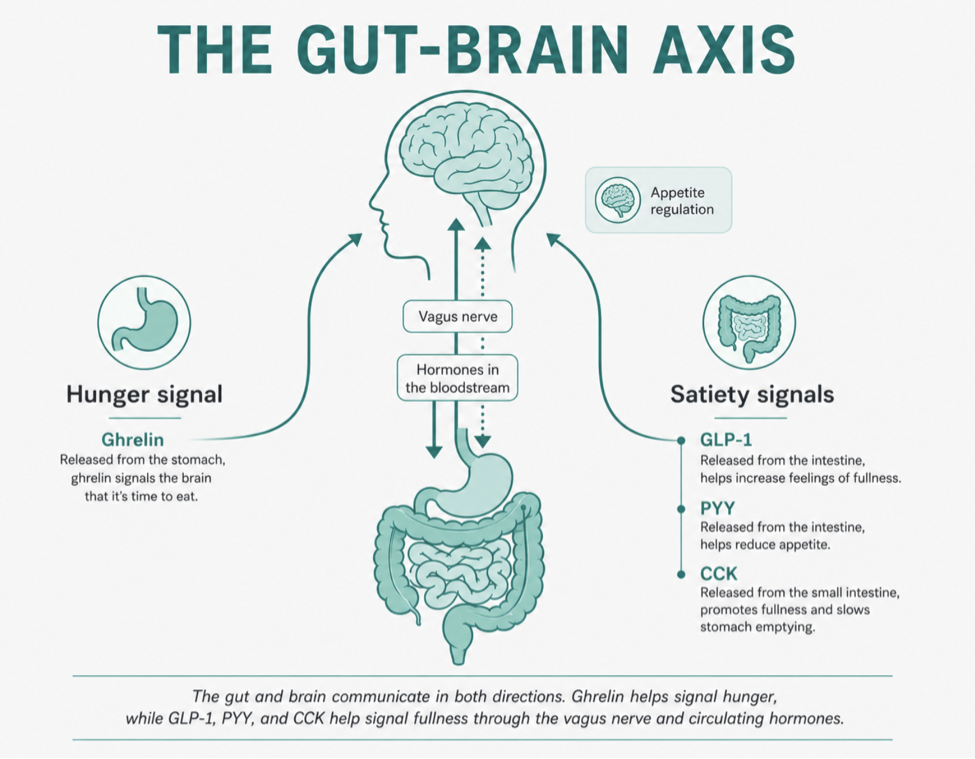
The gut-brain axis runs in both directions. Hunger and satiety signals travel between your digestive system and brain via both the vagus nerve and circulating hormones.
The four hormones that control how hungry you feel
Several hormones play key roles in appetite regulation, but four are particularly important for understanding why eating behaviour is so difficult to consciously control.
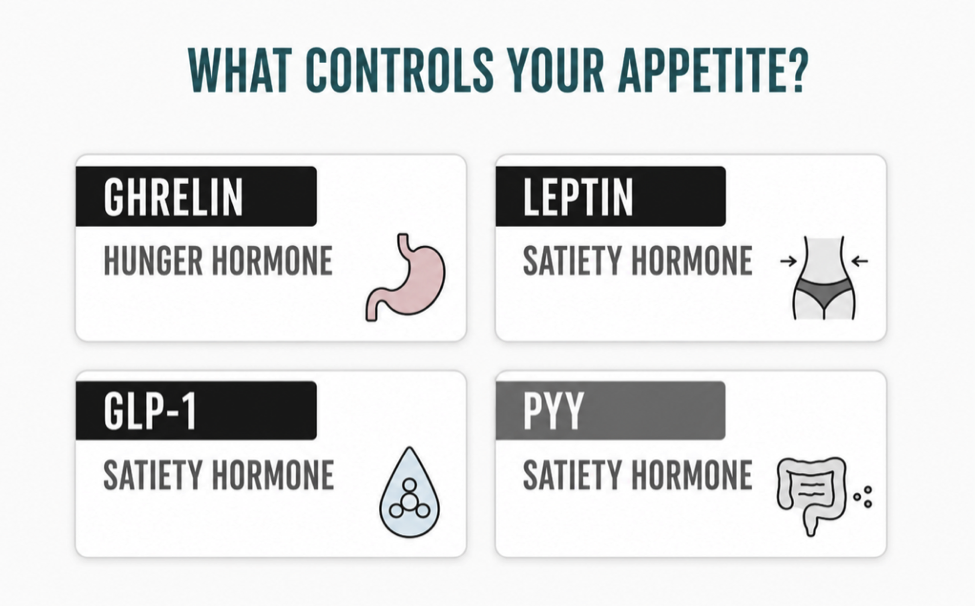
Four hormones dominate appetite regulation: ghrelin drives hunger, while leptin, GLP-1, and peptide YY signal fullness through different mechanisms and timescales.
Ghrelin: the hunger hormone
Ghrelin is produced primarily in the stomach and is the only known circulating hormone that actively stimulates appetite. Ghrelin levels rise before meals, peak when you are hungry, and fall after eating. They also rise significantly during calorie restriction, which is one of the core reasons dieting becomes harder over time: the longer you restrict your intake, the more your body pushes back with elevated hunger signals.
fMRI studies have shown that ghrelin administration increases activity in brain regions associated with reward and food motivation, meaning it does not just make you feel hungry in an abstract way. It makes food more appealing, more rewarding, and harder to resist.
| “ | Ghrelin levels remain elevated for at least 12 months after calorie-restricted weight loss, sustaining a heightened drive to eat long after the diet itself has ended. Sumithran et al., New England Journal of Medicine, 2011 |
Leptin: the fullness signal from fat
Leptin is produced by your fat cells and signals to the hypothalamus that energy stores are adequate, reducing appetite and increasing energy expenditure. In theory, this sounds like a good self-regulating system. In practice, it breaks down in people living with obesity.
When fat stores are large, leptin levels are high. But prolonged exposure to high leptin causes the brain to become leptin resistant: the hypothalamus stops responding effectively to the signal, in the same way that ears habituate to background noise. The result is a cruel paradox where people with obesity often have very high leptin levels but still feel persistently hungry because the brain has effectively stopped listening.
GLP-1 receptor agonists have been shown to partially restore leptin sensitivity, which is one of the reasons they outperform simple calorie restriction in appetite management.
GLP-1: your gut’s natural off switch
Glucagon-like peptide-1, or GLP-1, is a hormone secreted by specialised L-cells in your small intestine and colon when food is present. It does several things simultaneously: it slows gastric emptying (meaning food moves more slowly from your stomach into your intestine, prolonging the sensation of fullness), it stimulates insulin release, it suppresses glucagon, and critically, it sends satiety signals directly to the hypothalamus and brainstem via receptors in both organs.
GLP-1 also reduces the rewarding quality of food by acting on dopaminergic pathways in the brain. People on GLP-1 receptor agonists often describe not just feeling less hungry, but genuinely caring less about food. The urge to eat simply becomes quieter.
In people with obesity, GLP-1 secretion is often reduced and its effects blunted, contributing to impaired satiety signalling. This is not a character failing. It is a physiological feature of how the condition develops and maintains itself.
Peptide YY: the slow brake
Peptide YY (PYY) is released from the same intestinal L-cells as GLP-1, but its primary action is to slow gastric emptying and reduce appetite over a longer timeframe after eating. It works by inhibiting orexigenic (hunger-promoting) neurons and activating satiety pathways in the hypothalamus. Both PYY and GLP-1 levels are suppressed during calorie restriction, adding to the hormonal headwinds facing anyone trying to maintain weight loss through diet alone.
Why willpower alone cannot win this argument
The practical consequence of this hormonal system is that hunger and fullness are not primarily under voluntary control. Your prefrontal cortex, the part of the brain that makes deliberate decisions, is downstream of these hormonal signals. When ghrelin is high and leptin signalling is blunted, the experience of hunger is not a gentle reminder to eat. It is a persistent, biochemically generated drive that competes directly with conscious decision-making.
Research consistently shows that trying to suppress this drive through conscious restraint produces short-term results at best. The Minnesota Starvation Experiment, one of the most thorough studies of calorie restriction ever conducted, found that food preoccupation, cravings, and distress around eating intensified dramatically during restriction and took months to resolve after normal eating resumed. This was in healthy, motivated young men with no prior history of disordered eating.
| “ | When people attempt calorie restriction, ghrelin rises, leptin sensitivity falls, and resting metabolic rate decreases. All three changes persist after the diet ends, creating a sustained biological environment that favours weight regain. Hall et al., Obesity, 2016; Sumithran et al., NEJM, 2011 |
The willpower framing of weight management is not just ineffective. It is actively misleading, because it attributes a biological problem to a personal failure, which leads to shame rather than solutions.

Willpower-based approaches fight the biology of hunger. Hormone-based approaches work with it, addressing the root signals that drive appetite rather than asking for conscious restraint against them.
Where GLP-1 medications fit into this picture
Understanding the gut-brain axis makes the mechanism behind GLP-1 medications immediately logical. Rather than asking you to override your hunger signals through determination, these medications work with the same biological pathways that your body already uses to regulate appetite.
GLP-1 receptor agonists bind to the same receptors that your body’s naturally occurring GLP-1 uses. They slow gastric emptying, reduce ghrelin, enhance leptin signalling, and directly activate satiety centres in the hypothalamus and brainstem. The result is a reduction in the biological drive to eat, not just a conscious effort to eat less.
Mounjaro (tirzepatide) goes further still. It is a dual agonist that activates both GLP-1 receptors and GIP (glucose-dependent insulinotropic polypeptide) receptors. GIP has its own role in appetite regulation and fat metabolism, and the combined activation of both pathways produces weight loss outcomes that consistently exceed what GLP-1 activation alone achieves. In the SURMOUNT-1 clinical trial, tirzepatide produced up to 23% body weight loss, outperforming every prior pharmacological treatment.
That shift in thinking is why medically supported weight loss is increasingly framed around biology rather than willpower. For readers considering a clinical route, Voy provides access to Mounjaro treatment through a clinician-reviewed process, including an online assessment, dose planning, ongoing support, and delivery after approval.
The real-world outcomes when biology is addressed
When weight loss treatment addresses the hormonal drivers of appetite rather than just asking people to eat less, the outcomes are measurably different from what dieting alone produces.
Data published from Voy’s supervised programme, covering over 125,000 members, showed 24% average weight loss at 12 months, compared to 16% in controlled clinical trials of the same medication. The higher real-world figure reflects what happens when medication is combined with ongoing clinical monitoring, behavioural coaching, and dose adjustment over time.
The quality-of-life improvements go beyond weight itself. Binge eating scores improved in 94% of members. Depression scores improved in 74%. Both outcomes reflect what happens when the biological noise driving disordered eating is reduced: the psychological relationship with food often improves alongside the physical one.
| “ | 94% of Voy members showed improved binge eating scores during treatment. 74% showed improved depression scores. 100% reported improved quality of life at 12 months. Voy outcome data published in JMIR and Cureus, 2025 (N=125,000+). Individual results may vary. Treatment subject to clinical suitability. |
What this means practically
If you have struggled with hunger, cravings, or appetite that feels out of proportion to what you have eaten, the gut-brain axis science offers a more useful explanation than a lack of discipline. Your appetite is regulated by a sophisticated hormonal system that evolved to protect you from starvation, and when that system is disrupted by obesity, hormonal imbalance, or chronic calorie restriction, the experience of hunger becomes genuinely harder to manage than it is for people whose systems are working normally.
The approaches most likely to produce lasting results are those that address these underlying hormonal mechanisms rather than asking you to override them indefinitely. That includes:
- Eating patterns that support GLP-1 and PYY release, including adequate protein and fibre at meals
- Sleep and stress management, both of which affect ghrelin and cortisol
- For those who need additional support, medically supervised treatment that works directly with the appetite hormones driving hunger
Understanding that hunger is a biological signal, not a moral test, is the first step toward finding a strategy that actually works with your body.
Frequently asked questions
What is the gut-brain axis?
The gut-brain axis is the bidirectional communication system between your gastrointestinal tract and your brain. It operates through both the vagus nerve (direct neural signals) and circulating hormones produced by your gut, fat tissue, and pancreas. This system regulates appetite, digestion, energy balance, and even mood.
What is ghrelin and why does it matter for weight loss?
Ghrelin is the main hunger hormone, produced in the stomach when it is empty. Levels rise before meals and during calorie restriction. Research shows ghrelin remains elevated for at least a year after dieting, which is one of the key biological reasons weight regain is so common. GLP-1 medications reduce ghrelin levels, which is part of why they reduce hunger more effectively than diet restriction alone.
What does GLP-1 do naturally in the body?
GLP-1 is a satiety hormone produced in your small intestine in response to food. It slows gastric emptying, stimulates insulin release, suppresses appetite signals in the hypothalamus, and reduces the rewarding quality of food by acting on dopaminergic pathways. In people with obesity, natural GLP-1 signalling is often impaired, meaning satiety signals are weaker than normal.
Why is willpower not enough for long-term weight loss?
Because appetite is primarily regulated by hormonal signals that operate below conscious awareness. When ghrelin is elevated and leptin signalling is blunted, as they both are during and after calorie restriction, the experience of hunger is a sustained biological drive that competes directly with deliberate decision-making. Conscious restraint can override these signals temporarily, but not indefinitely. Approaches that address the underlying hormones produce more durable outcomes.
How does Mounjaro affect appetite hormones?
Mounjaro (tirzepatide) is a dual GLP-1 and GIP receptor agonist. It activates the same receptors your body’s natural GLP-1 uses to signal fullness, while also activating GIP receptors involved in fat metabolism. This reduces ghrelin, enhances leptin sensitivity, slows gastric emptying, and directly activates satiety centres in the brain, addressing multiple points in the gut-brain appetite system simultaneously.
| This article is for informational purposes and does not constitute medical advice. Always consult a healthcare professional before starting any weight loss treatment. Individual results may vary. Treatment is subject to clinical suitability. Results achieved alongside a reduced-calorie diet and increased physical activity. |
Sources
• Barakat GM et al. Satiety Homeostasis: The Role of GLP-1, Gut Microbiome and Neurotransmitters. Journal of Physiological Sciences, 2024.
• Tsoukas MA et al. Mechanisms of GLP-1 Receptor Agonist-Induced Weight Loss. The American Journal of Medicine, 2025.
• Clarke et al. The gut-brain axis in appetite, satiety, food intake, and eating behavior. Pharmacology Research and Perspectives, 2024.
• Balonov I et al. Satiety Regulation in Obesity. Biomedical Journal of Scientific and Technical Research, 2025.
• Sumithran P et al. Long-Term Persistence of Hormonal Adaptations to Weight Loss. New England Journal of Medicine, 2011.
• Hall KD et al. Persistent metabolic adaptation 6 years after The Biggest Loser competition. Obesity, 2016.
• Blundell J et al. Effects of once-weekly semaglutide on appetite, energy intake, food preference, meal size and appetite control. Diabetes, Obesity and Metabolism, 2017.
• Voy outcome data: JMIR, 2025 (N=125,000+). Cureus, 2025. Diabetes Obesity and Metabolism, 2025.
Other Articles You May Find of Interest...
- How to Build Home Exercise Programs Patients Actually Follow
- How to Choose the Right Strength Training Program in Dallas
- Best Bird-Friendly Coffee Brands (2026): Shade-Grown, Sustainable Options
- Prozac: Does It Cause Weight Gain or Loss?
- Building a Stronger You: Smart Strategies for a Balanced Fitness Routine
- The Mounjaro Transition: How To Move From Medication-Led Weight Loss to Long-Term Control
- Does Flomax Contribute to Weight Gain? Uncovering the Truth














